I. Environment rapidly upregulates serotonin 2A receptor expression via immediate early gene Egr3
Our lab has discovered that levels of serotonin (5-HT) 2A receptors can be rapidly upregulated in the frontal cortex by a brief environmental intervention. Just 6 to 8 hours of sleep deprivation significantly increases expression of the 5-HT2A receptor gene (Htr2a) and membrane-bound receptor protein in the frontal cortex of mice. We found that this requires the activity-dependent immediate early gene Egr3. Moreover, EGR3 protein binds to the promoter of the 5-HT2A gene in vivo, and drives expression of reporter promoter-linked reporter constructs in vitro, suggesting that EGR3 directly regulates the 5-HT2A in response to environmental stimuli. https://www.biorxiv.org/content/10.1101/634410v1
Ongoing work in the lab is investigating the cortical layers in which this occurs, and how this may affect behaviors that are influenced by both Egr3 and 5-HT2A receptors.
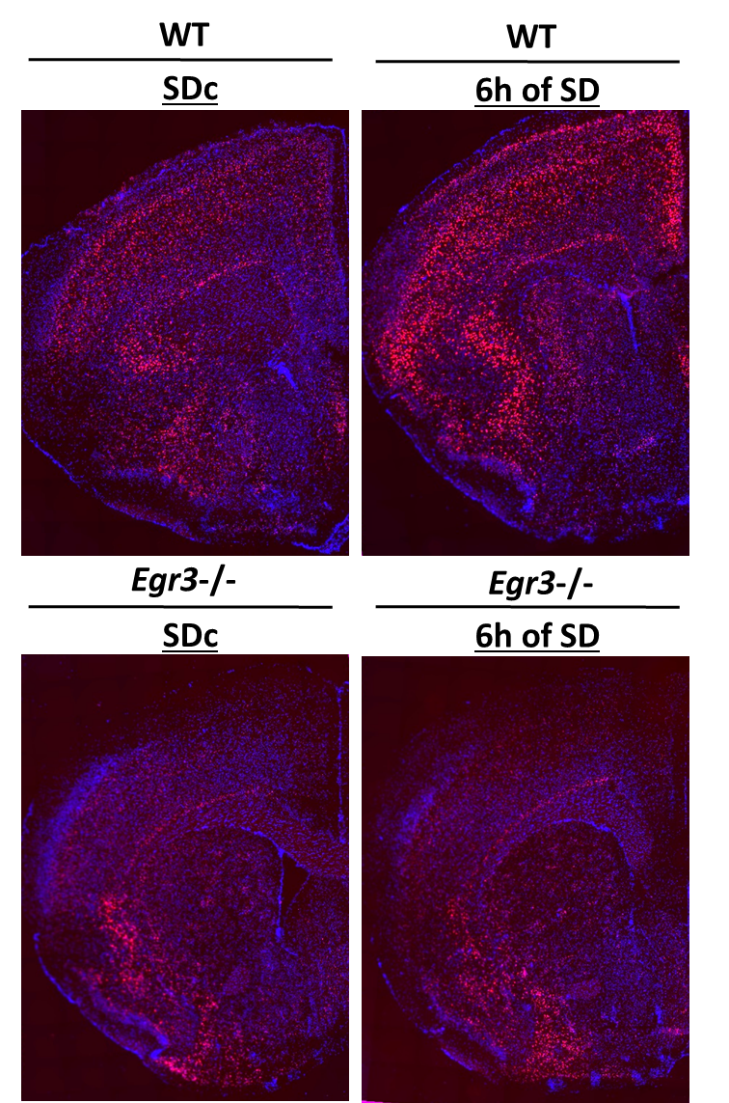
Sleep deprivation upregulates cortical Htr2a expression in an Egr3-dependent manner.
Brain sections from wildtype (WT) and Egr3-/- mice show the significant upregulation of Htr2a mRNA (in red) in WT mice following 6 hours of sleep deprivation (SD), compared with control sleeping animals (SDc). Significantly lower Htr2a levels in Egr3-/- mice (SDc) do not increase following SD. Method: RNAscope in situ hybridization, nuclei are visible with blue DAPI staining. https://www.biorxiv.org/content/10.1101/634410v1
II. Molecular Mechanisms Underlying the Therapeutic Effect of ECT
The most effective treatment identified to date for severe mood and psychotic disorders is electroconvulsive therapy (ECT). These mental illnesses, which include major depression, with or without psychosis, bipolar disorder, and schizoaffective disorder, affect upwards of 6.5% of adults in the U.S. and account for a major proportion of disability and mortality in society.
Unfortunately, despite nearly 80 years of safe use, the negative image of ECT drastically limits its implementation. Discovery of the mechanisms that are responsible for the therapeutic effect of ECT would permit the development of next-generation treatments to achieve the same superior result without seizure.
This project investigates the hypothesis that the immediate early gene transcription factor Egr3, which is induced by ECT and regulates growth factor expression and neuronal plasticity, is a critical mediator of this highly effective treatment for severe mental illness.
One of the findings of this study is that Egr3 is required for induction of BDNF expression in the hippocampus by electroconvulsive seizure (the same process used for ECT in humans).
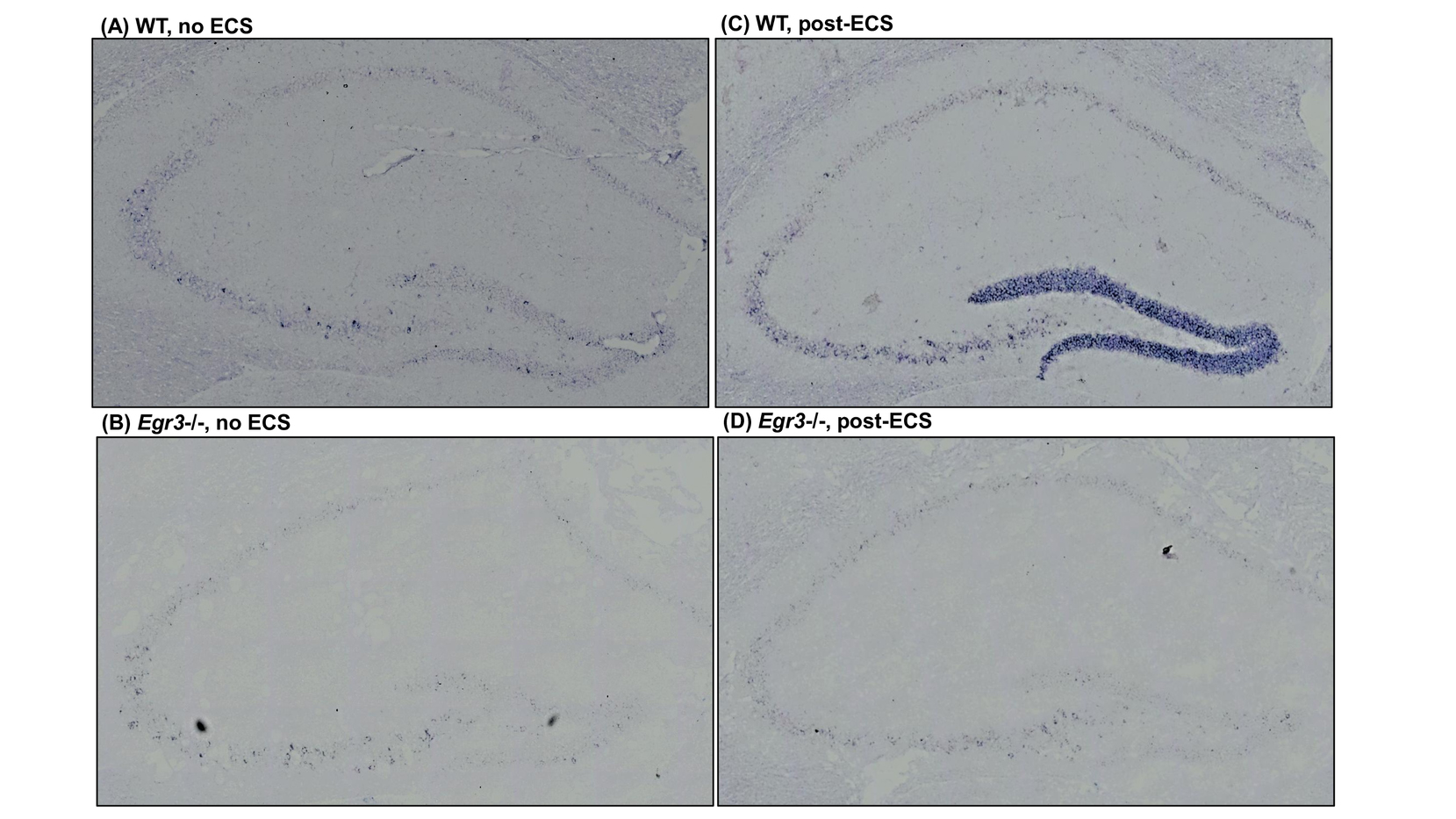
Egr3 is required for induction of hippocampal Bdnf by electronconvulsive seizure (ECS).
In situ hybridization labeling of Bdnf mRNA in the hippocampus of WT and Egr3–/– mice at baseline (A,B) and 1 h following ECS (C,D). ECS induces high-level Bdnf expression in the dentate gyrus of WT mice (A vs. C), which fails to occur in Egr3–/– mice (B vs. D). https://www.frontiersin.org/articles/10.3389/fnbeh.2018.00092/full

